Stille Cross-Coupling Reactions of Alkenylstannanes with Alkenyl Iodides Mediated by Copper(I) Thiophene-2-carboxylate: A Density Functional Study | Organometallics

Stille Cross-Coupling Reactions of Alkenylstannanes with Alkenyl Iodides Mediated by Copper(I) Thiophene-2-carboxylate: A Density Functional Study | Organometallics
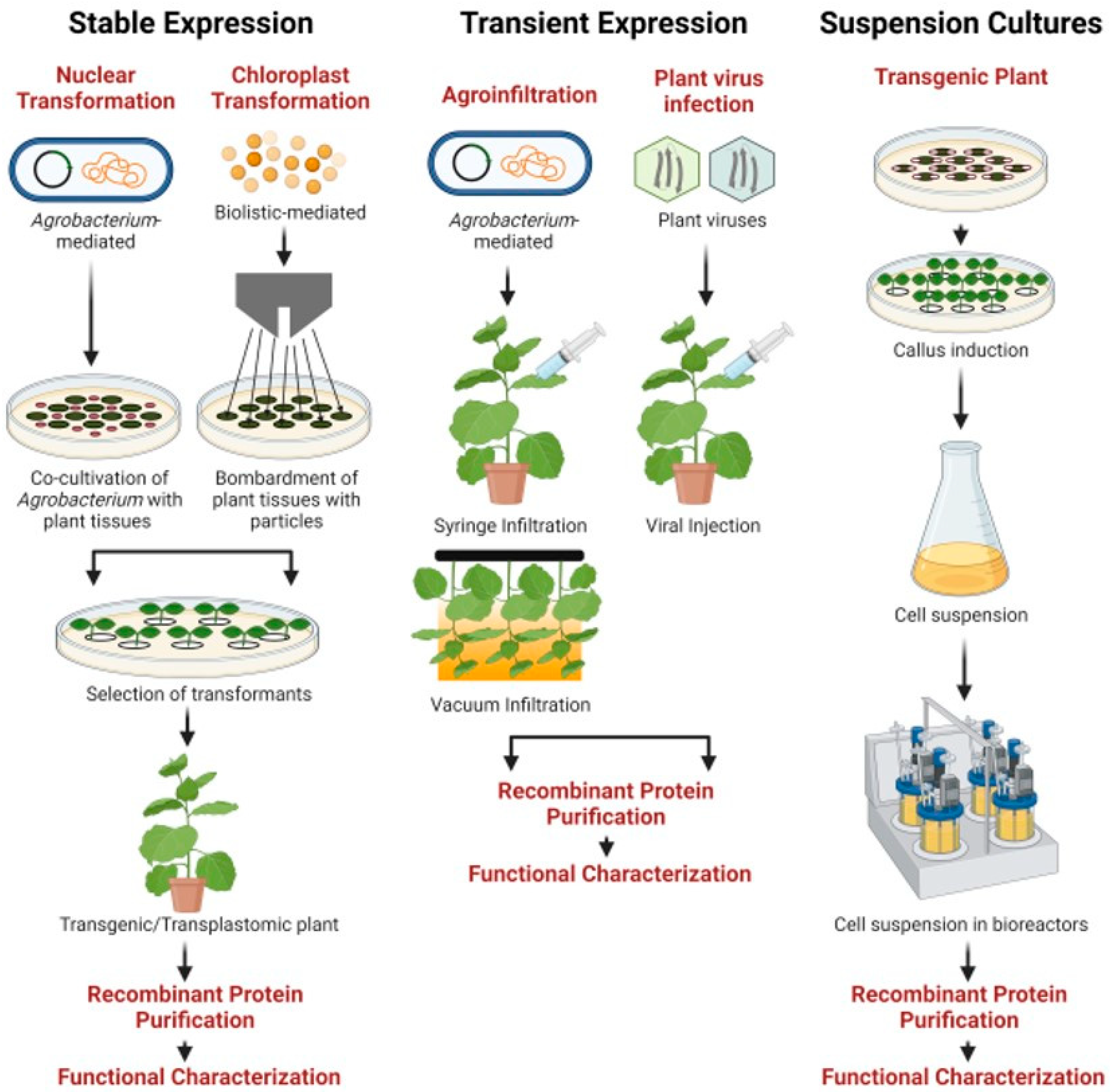
Pathogens | Free Full-Text | Potential for Developing Plant-Derived Candidate Vaccines and Biologics against Emerging Coronavirus Infections
Papers Past | Parliamentary Papers | Appendix to the Journals of the House of Representatives | 1936 Session I | DEPARTMENT OF INDUSTRIES AND COMMERCE (NINETEENTH...

Counterintuitive Lanthanide Hydrolysis-Induced Assembly Mechanism | Journal of the American Chemical Society
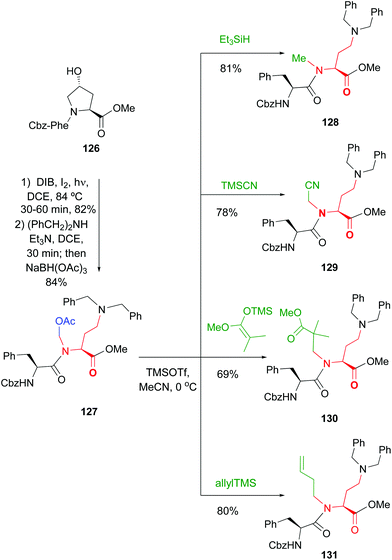
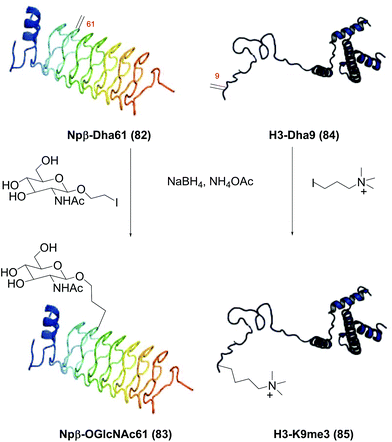
Site-selective modification of peptide backbones - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00892G

Protective effect of ascorbic acid on cadmium-induced hypertension and vascular dysfunction in mice | SpringerLink
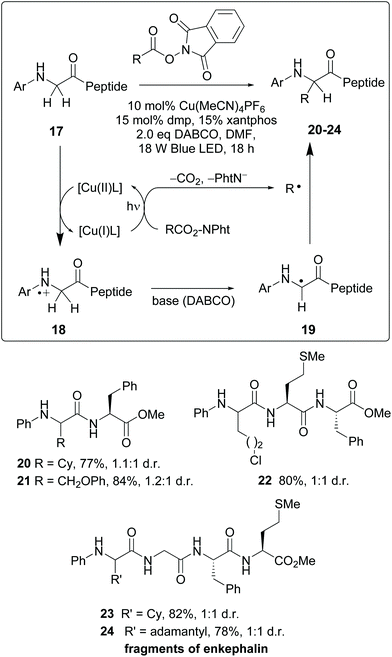
Site-selective modification of peptide backbones - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00892G

Stille Cross-Coupling Reactions of Alkenylstannanes with Alkenyl Iodides Mediated by Copper(I) Thiophene-2-carboxylate: A Density Functional Study | Organometallics
![Conformational Preferences of Allene Ketones in Lewis Base Catalysis: Synthesis of 4H‐Pyrans and 3,4‐Dihydro‐2H‐pyrans via α‐Regioselective [4+2] Annulations of γ‐Substituted Allene Ketones and Activated Alkenes - Hou - 2021 - Asian Journal Conformational Preferences of Allene Ketones in Lewis Base Catalysis: Synthesis of 4H‐Pyrans and 3,4‐Dihydro‐2H‐pyrans via α‐Regioselective [4+2] Annulations of γ‐Substituted Allene Ketones and Activated Alkenes - Hou - 2021 - Asian Journal](https://onlinelibrary.wiley.com/cms/asset/8d23e080-3bcc-4ee0-98b8-d58b2594f085/ajoc202100523-toc-0001-m.jpg)
Conformational Preferences of Allene Ketones in Lewis Base Catalysis: Synthesis of 4H‐Pyrans and 3,4‐Dihydro‐2H‐pyrans via α‐Regioselective [4+2] Annulations of γ‐Substituted Allene Ketones and Activated Alkenes - Hou - 2021 - Asian Journal